The Roman architect Vitruvius’ book “De Architectura” heavily influenced the building materials and techniques used by architects and artisans during the Venetian Republic. The Roman art of building stemmed from Greek culture, and the construction techniques and materials originated from the Egyptians and the Persians. This knowledge was gradually transmitted through oriental culture (primarily from East Iran) via the Mesopotamian and Mediterranean civilizations.
Lime plaster has been used in construction since around 7500 BC, when the people of ‘Ain Ghazal in Jordan used it for their walls. Ancient civilizations, including the Greeks, Egyptians, Mayas, Incas, as well as the early Chinese and Indian dynasties, incorporated lime into their architectural masterpieces. The Egyptians coated the walls of their tombs with lime and gypsum plaster, while the Romans used lime-sand mixtures. After the fall of the Roman Empire, marble dust was added to plaster, and the Italians and French later developed stucco (lime plaster) techniques.


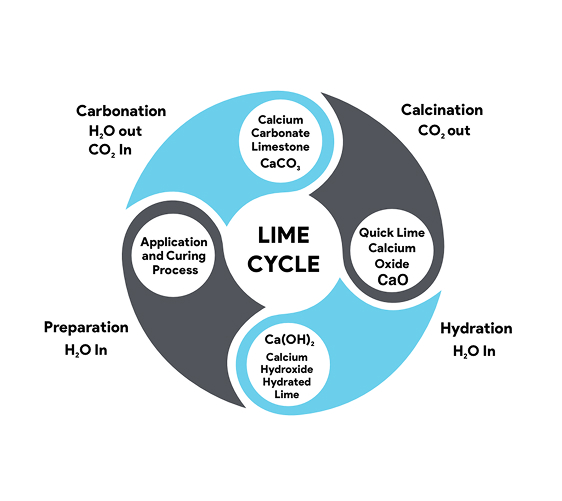
Lime plaster is created through a series of chemical reactions and processes involving calcium carbonate. The primary ingredient, calcium oxide (commonly known as lime), is derived from heating calcium carbonate sources such as limestone, marble, chalk, or shells in a kiln at temperatures ranging from 900°C to 1200°C. The heating process, known as calcination, drives off carbon dioxide (CO2), leaving behind anhydrous calcium oxide, also referred to as quicklime.
Quicklime, a highly reactive and caustic substance, undergoes a significant transformation when it comes into contact with water. This process, called slaking, involves soaking the quicklime in water, which causes it to react and change into calcium hydroxide, also known as slaked lime or hydrated lime.
The slaked lime, once dried and ground into a fine powder, is referred to as hydrated lime. The powdered form is the key ingredient in lime plaster and is mixed with water and aggregates like sand to create a workable paste. Once it has been applied, the lime plaster undergoes a process called carbonation, where it sequesters the surrounding carbon dioxide that is present in the air to then slowly harden and revert back to calcium carbonate or limestone. This carbonation process is essential for the durability and longevity of lime plaster, as it allows the material to strengthen over time, creating a breathable and resilient mineral skin.
There are two main types of lime: air lime and hydraulic lime.
Air lime is produced by heating calcareous stones containing less than 5% clay matter. It can be categorized based on purity: “fat lime” (over 95% carbonates) and “lean lime” (up to 5% clay). It can exist in either a raw state (calcium oxide or quicklime) or a hydrated state (calcium hydroxide), and its setting agent is carbon dioxide.
Obtained when the stone is rich in clay, iron, and alumina. The setting agents are both water and carbon dioxide. Natural Hydraulic Lime (NHL) is classified for different uses:
| TYPE OF HYDRAULIC LIME | CLAY CONTENT | USES |
|---|---|---|
| Feebly hydraulic NHL 2 | <8% | Interiors and Exteriors |
| Moderately hydraulic NHL 3.5 | 8—19% | Most exterior areas |
| Eminently hydraulic NHL 5 | >20% | Harsh environments |

